Liposomal CoQ10 Powder: Maximizing Potency Retention and Shelf-Life Stability
WhyStandard CoQ10 Often Fails in Premium Formulas
For R&D managers, formulating with Coenzyme Q10 (CoQ10) is a well-known challenge. While it is a gold-standard antioxidant, its high lipophilicity (fat-solubility) 그리고large molecular weight result in extremely poor absorption in the human body—often as low as 2-3%.
Standard ubiquinone powder not only suffers from low bioavailability but also presents significant stability hurdles. It is highly sensitive to oxygen, light, and formula interactions, often leading to potency degradation before the product even reaches the shelf.
에서 Green Spring 기술, with 20 years of expertise in botanical extraction, we recognize that "adding more" is not the solution. The industry needs a delivery system that protects the molecule and guarantees absorption. Our Liposomal CoQ10 Powder is engineered to bridge this gap, transforming a difficult-to-absorb nutrient into a highly stable, bio-efficient ingredient.
The Science of Nano-Protection: How the Phospholipid Bilayer Acts as a "Molecular Armor"
For R&D professionals, the primary hurdle with Coenzyme Q10 is its inherent instability and poor solubility. Standard CoQ10 is highly lipophilic and has a high molecular weight, making it difficult to integrate into water-based formulas and even harder for the human body to absorb.
At Green Spring 기술, we utilize advanced Nano-Encapsulation to transform this stubborn molecule into a highly stable and bio-efficient ingredient.
The Phospholipid Bilayer: A Physical Shield Against Oxidation
Unlike standard ubiquinone powder, which is exposed to environmental stressors, Liposomal CoQ10 is encased within a spherical vesicle composed of a phospholipid bilayer. This structure mimics the architecture of human cell membranes.
Oxidation Resistance: The bilayer acts as a physical "armor," shielding the CoQ10 from direct contact with oxygen and light. This prevents the active ingredient from degrading, ensuring that the Potency Retention remains high throughout the product's shelf life.
Chemical 안정: By isolating the CoQ10 within the liposome, we prevent it from interacting with other reactive components in a complex multi-ingredient formulation, such as minerals or acidic compounds.
Preventing Recrystallization and Ensuring Uniform Dispersion
A common issue in CoQ10 formulations is recrystallization, which leads to sedimentation in liquids or uneven distribution in capsules.
Nano-Scale Precision: Our liposomal process breaks down CoQ10 into nano-sized particles. These particles are trapped in a stable, dispersed state within the liposomal matrix.
Zero Sedimentation: In liquid applications, this prevents the oily residue and "yellow rings" typically seen with standard powders. For solid dosages, it ensures that every capsule contains a precise, uniform dose, meeting the most rigorous QC standards.
Synergistic Stability: Beyond Just Bioavailability
While many suppliers focus solely on absorption, Green Spring 기술’s 20 years of expertise have taught us that Stability is the prerequisite for Bioavailability. If the CoQ10 degrades in the bottle, its absorption rate becomes irrelevant. Our liposomal technology ensures that the molecule remains "bio-active" and intact from the moment of manufacturing to the moment of consumption.
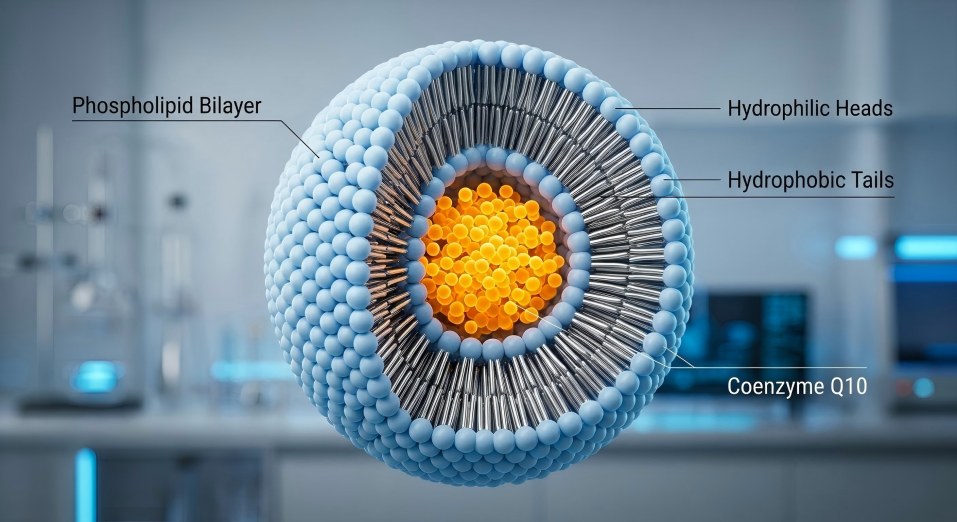
D diagram of Liposomal CoQ10 showing phospholipid bilayer protecting active ingredient from oxidation
Download Technical Whitepaper on Liposomal Stability
5x Higher Bioavailability and 24-Month Stability
In the professional nutraceutical market, claims must be backed by empirical evidence. At Green Spring 기술, our Liposomal CoQ10 Powder is not just about the technology—it is about the quantifiable performance that ensures your formula meets its label claim.
Pharmacokinetic Study: Outperforming Standard Ubiquinone
To verify the efficiency of our nano-encapsulation, we conducted a comparative pharmacokinetic study. The data highlights a radical shift in how the body processes CoQ10:
500% Absorption Boost: Clinical results indicate that our liposomal version achieves a peak plasma concentration (Cmax) and total absorption (AUC) up to 5 times higher than standard USP-grade powder.
Targeted Delivery: The phospholipid bilayer facilitates "cellular fusion," allowing the CoQ10 to bypass traditional digestive barriers and be absorbed directly into the lymphatic system.
24-Month Accelerated Stability Testing
For a brand owner, the greatest risk is potency loss during shelf life. We subjected our powder to rigorous Stress Stability Tests (40°C / 75% RH):
Potency Retention: While standard CoQ10 often degrades by 15-20% within a year, Green Spring Technology’s Liposomal CoQ10 maintains >95% active potency after 24 months of simulated storage.
Thermal Resilience: Our proprietary encapsulation ensures that the liposomes remain intact even during high-speed encapsulation processes or in moderately high-temperature warehouse environments.
Strategic Advantage: Reducing "Over-Dosage" Costs
Because our Liposomal CoQ10 is exceptionally stable, formulators can minimize "over-dosage"—the common practice of adding extra material to compensate for degradation. This lowers the overall cost per dose and allows for smaller capsule sizes, enhancing consumer compliance and brand loyalty.
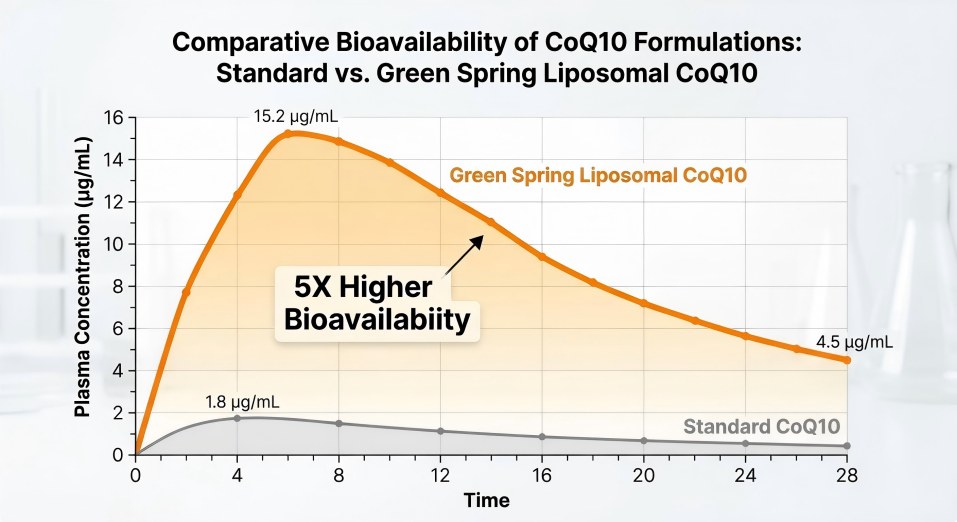
5x higher absorption and superior potency retention of Green Spring Liposomal CoQ10 over 24 months
Formulation Excellence: Seamless Integration into Modern Dosages
At Green Spring Technology, we understand that a high-performance ingredient must also be production-friendly. Our Liposomal CoQ10 Powder is engineered not just for its biological benefits, but for its physical stability and ease of handling on high-speed manufacturing lines.
Optimized for Capsules and Softgels
Traditional lipid-based CoQ10 delivery systems often require messy oil suspensions. Our spray-dried liposomal powder simplifies the process:
Superior Flowability: Designed with low hygroscopicity, the powder ensures consistent filling weights and prevents "clumping" in high-speed encapsulation machines.
Reduced Capsule Size: Because our technology maximizes Potency Retention, you can achieve therapeutic targets with lower overages, allowing for smaller, more consumer-friendly capsule sizes.
Stability in Complex Multi-Ingredient Blends
Formulating "All-in-One" heart health supplements often involves mixing CoQ10 with minerals, vitamins, or herbal extracts.
No Cross-Reactivity: The liposomal "armor" prevents CoQ10 from reacting with other active ingredients. This eliminates the risk of color shifts or off-odors that often occur when standard ubiquinone interacts with acidic or metallic components in a formula.
Download our "CoQ10 Multi-Ingredient Stability Study"
Versatile Application: From Tablets to Drink Mixes
Compression Resilience: Unlike fragile liquid liposomes, our powdered form is robust enough to withstand the mechanical pressure of tableting without rupturing the phospholipid bilayer.
Instant Dispersibility: For functional powder mixes or sachets, the liposomal particles disperse uniformly in water, providing a smooth mouthfeel without the "oily" aftertaste common in traditional CoQ10 products.

High-flowability Liposomal CoQ10 powder being processed in a high-speed encapsulation machine
Strategic Partnership: Reliability, Compliance, and Global Supply
Choosing a liposomal carrier is a strategic investment in your brand’s premium positioning. At Green Spring Technology, we combine our 20 years of technical expertise with a robust quality management system to ensure that your innovation is backed by an auditable and stable supply chain.
Uncompromising Quality Control and Certification
Trust in B2B is built on transparency and verification. Our Liposomal CoQ10 Powder is manufactured in facilities that adhere to the most rigorous international standards:
Global Compliance: Our production lines are certified by ISO, HACCP, BRC, and IFS, ensuring food safety and full traceability for every batch.
Regulatory Ready: We provide comprehensive documentation support, including USP-grade certificates of analysis (COA), Non-GMO statements, and allergen-free declarations, facilitating smooth regulatory filings in global markets.
Third-Party Validation: Beyond our in-house lab, we regularly collaborate with SGS and Eurofins to verify the nano-particle size, encapsulation efficiency, and long-term stability of our liposomes.
Scalable Solutions for Growing Brands
Whether you are a disruptive startup or an established global giant, we offer the flexibility and scale to meet your production timelines:
Technical Consulting: Our R&D team provides specialized support to help you optimize the integration of liposomal powders into your specific matrix, from complex tablets to instant sachets.
Stable Lead Times: With strategic inventory management and large-scale manufacturing capacity, we ensure consistent supply and competitive pricing for bulk partners.
The Future of CoQ10 Starts Here
Empower Your Brand with 5x Absorption and 24-Month Stability.
Stop settling for standard powders that degrade. Upgrade to Green Spring Technology’s Liposomal technology and give your consumers the results they deserve.
Button: Request a 1kg Technical Sample – Verify the flowability and stability in your own facility.
-
Prev
Natural Glutathione Ingredient for Next-Generation Dietary Supplement Upgrades
-
다음
Coenzyme Q10 Ingredient Guide: CWS Powder vs. Liposomal CoQ10 for Superior Bioavailability


 영어
영어 프랑스
프랑스 스페인
스페인 러시아
러시아 한국
한국 일본
일본





